- Clinically meaningful improvement of 25 ETDRS letters equivalent versus nadir of BCVA in GS010-treated eyes at 96 weeks
- Results coherent with those of REVERSE: durable bilateral improvement in vision, despite the intervening acute phase in RESCUE
- Recovery in BCVA greatly exceeds results from published natural history studies
- GenSight Biologics now expects to share these findings with regulatory agencies and file for approval in Europe in Q3 2020
Paris, France, Monday, September 23, 2019, 7.30 am CEST – GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on discovering and developing innovative gene therapies for retinal neurodegenerative diseases and central nervous system disorders, today reported the first set of results from Week 96 of the RESCUE Phase III clinical trial. The trial evaluated the efficacy and safety of a single intravitreal injection of GS010 (rAAV2/2-ND4) in 39 subjects whose visual loss due to 11778-ND4 Leber Hereditary Optic Neuropathy (LHON) commenced up to 6 months prior to study treatment. Week 96, which marks the time when individual patient profiles can be analyzed, is the last of the scheduled readouts for the RESCUE trial and completes the data collection from GS010’s pivotal trials in Europe.
The results point to continued efficacy of GS010 two years past injection, with best-corrected visual acuity (BCVA) sustaining a clinically meaningful improvement over nadir. Having been treated early in the course of the disease, RESCUE patients’ vision initially deteriorated to a worst point, or nadir, before beginning to recover.
Figure 1: Time Course of Best-Corrected Visual Acuity (BCVA) in LogMAR
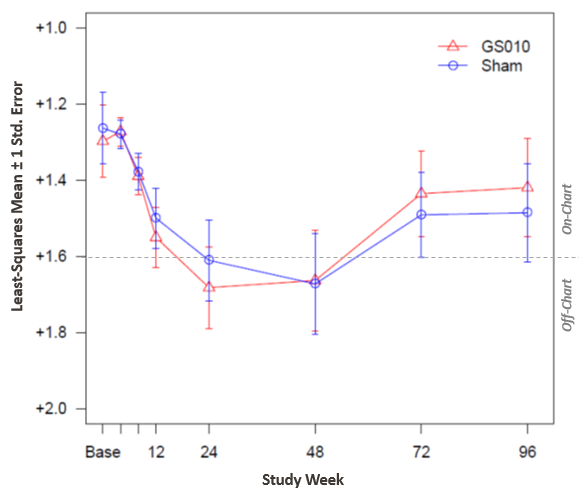
Note: A mixed model of analysis of covariance (ANCOVA) was used with change from baseline as the response, and subject, eyes of the subject as random factor, treatment and the baseline LogMAR value as covariates in the model.
When visual acuity is measured from the post-baseline nadir, the visual acuity of GS010-treated eyes in fact recovered significantly from the worst BCVA reading post-baseline.
Table 1: BCVA, Change from Nadir*, RESCUE
| Week 48 | Week 72 | Week 96 | ||||
| N | Mean (SD) | N | Mean (SD) | N | Mean (SD) | |
| All-GS010 Eyes | 36 | +12.8 (17.9) | 34 | +20.6 (26.3) | 34 | +24.9 (3.8) |
| All-Sham Eyes | 36 | +11.8 (15.6) | 33 | +21.7 (25.1) | 34 | +22.3 (3.8) |
Note: *”Nadir” defined as worst BCVA measured in LogMAR after baseline, up to the week of interest; baseline reading was excluded from consideration. Mean change was calculated using observed values (no data imputation).
GS010-treated eyes regained more than two-thirds of the initial loss occurring in the most acute phase of the disease. This improvement from nadir (-0.498 LogMAR mean improvement, or +24.9 ETDRS letters equivalent) corresponds to 5 lines of Snellen acuity and is far above the 3-line threshold commonly accepted as a clinically meaningful level of visual improvement. Such recovery of vision is unprecedented in any gene therapy trial. Moreover, these results demonstrate the durability of improvement seen in earlier readouts of this trial.
Vision in sham-treated eyes evolved in parallel fashion, continuing the bilateral improvement already observed in earlier Phase III readouts. The picture that has emerged is one of durable, bilateral recovery from the effects of the acute phase in LHON.
The BCVA results from RESCUE show a remarkable correspondence with those from the REVERSE trial, which studied the treatment of subjects at 6 to 12 months after onset of vision loss.
Figure 2: Time Course of Best-Corrected Visual Acuity (BCVA) in LogMAR, RESCUE and REVERSE Trials
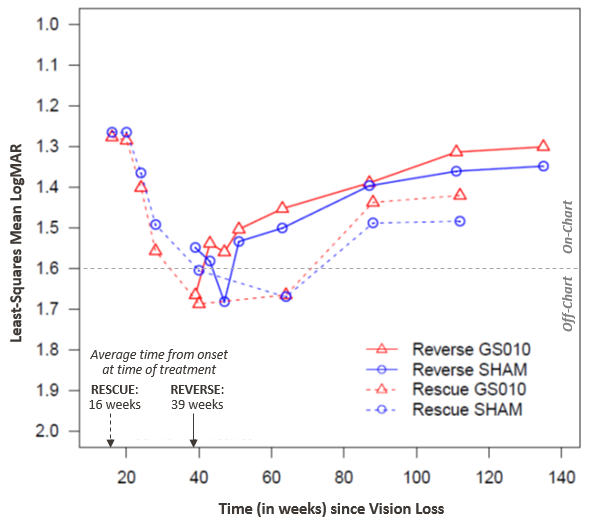
The visual evolution in RESCUE appears to be a phase shift of the REVERSE curves, with an additional impact from the acute phase. This coherence will be more rigorously explored in a meta-analysis of the pooled data from the two trials, which is planned for this year.
In previous analyses of these trials, LHON clinical experts confirmed that the visual recovery seen in these trials are at odds with and much superior to their observations from clinical practice. That natural history of vision in untreated LHON patients stands in strong contrast to acuities seen in both RESCUE and REVERSE.
A natural history study conducted by Santhera1 provides another way of assessing the results in RESCUE. In that study, 28% of subjects who had the 11778A mutation achieved the following definition of spontaneous “clinically relevant recovery” (CRR) from nadir in at least one eye:
- Improved by at least 10 ETDRS letters from nadir (for on-chart eyes at nadir), or
- Improved from an off-chart level of visual acuity at nadir to being able to read at least 5 ETDRS letters
By comparison, 58% of RESCUE subjects achieved this definition of CRR in at least one eye at Week 96, with GS010-treated eyes as likely to achieve this as sham-treated eyes (58% vs. 45%, p = 0.0956).
“The results from the RESCUE study are encouraging and convincing, particularly because we are seeing a similar pattern to the REVERSE study results,” said Dr. Mark L. Moster, Neuro-Ophthalmology, Wills Eye Hospital, Professor of Neurology and Ophthalmology at Thomas Jefferson University, Philadelphia, PA, and Principal Investigator in the RESCUE and REVERSE trials. “Patients in RESCUE were treated before the nadir so, as expected, they continued to worsen early on. But then from week 48 until week 96 they experienced a recovery from the nadir. That is much better than the natural history in any prior studies.”
Examination of other visual functions and biomarkers, including Contrast Sensitivity, show that these measures stabilized at Week 96. Based on preliminary analysis of the safety data, GS010 was well-tolerated after 96 weeks. There were no ocular serious adverse events or discontinuations that were due to ocular events. The ocular adverse events most frequently reported in the therapy group were mainly related to the injection procedure, except for the occurrence of intraocular inflammation (accompanied by elevation of intraocular pressure in some patients) that is likely related to GS010, and which was responsive to conventional treatment and without sequelae. There were no systemic serious adverse events or discontinuations that were related to study treatment or study procedure.
“These results are remarkable, showing the durable difference that GS010 can make for patients who would otherwise go blind due to the onset of LHON,” commented Bernard Gilly, Co-founder and Chief Executive Officer of GenSight. “These findings, which we will be discussing at the meetings we have planned with regulatory authorities, form a compelling core for the clinical and non-clinical data that support our marketing authorization application in Europe. GenSight is excited to have reached this milestone in GS010’s clinical development and energized by the prospect of pulling it all together for our European dossier.”
GenSight is planning to schedule a pre-submission meeting with the EMA in early 2020 and expects to submit application for marketing approval in Europe in the third quarter of 2020.
An End of Phase II meeting with the U.S. Food and Drug Administration (FDA) has been requested and is expected for November 2019.
GenSight will host a conference call today, September 23, 2019, at 10:30am CEST in French, and at 2.30pm CEST (8.30am EST) in English, to discuss in greater detail these results and the roadmap to submission.
Webcast & Conference call in French
Dial-in numbers:
- United States: +1 212 999 6659
- France: +33 (0) 1 7037 7166
- United Kingdom: +44 (0) 20 3003 2666
- Password: GenSight
Webcast link: https://channel.royalcast.com/webcast/gensightbiologicsfr/20190923_1/
Webcast & Conference call in English
Dial-in numbers:
- United States: +1 212 999 6659
- France: +33 (0) 1 7037 7166
- United Kingdom: +44 (0) 20 3003 2666
- Password: GenSight
Webcast link: https://channel.royalcast.com/webcast/gensightbiologicsen/20190923_1/
A replay of the calls and webcasts will be available by using the above links.
Reference:
- Magda et al (2019), “Natural History of Leber’s Hereditary Optic Neuropathy (LHON): Findings from a Large Patient Cohort”, Poster presented at NANOS March 16-21, 2019; Poster Session II: Scientific Advancements; Poster: 163
About GenSight Biologics
GenSight Biologics S.A. is a clinical-stage biopharma company focused on discovering and developing innovative gene therapies for retinal neurodegenerative diseases and central nervous system disorders. GenSight Biologics’ pipeline leverages two core technology platforms, the Mitochondrial Targeting Sequence (MTS) and optogenetics, to help preserve or restore vision in patients suffering from blinding retinal diseases. GenSight Biologics’ lead product candidate, GS010, is in Phase III trials in Leber Hereditary Optic Neuropathy (LHON), a rare mitochondrial disease that leads to irreversible blindness in teens and young adults. Using its gene therapy-based approach, GenSight Biologics’ product candidates are designed to be administered in a single treatment to each eye by intravitreal injection to offer patients a sustainable functional visual recovery.
About GS010
GS010 targets Leber Hereditary Optic Neuropathy (LHON) by leveraging a mitochondrial targeting sequence (MTS) proprietary technology platform, arising from research conducted at the Institut de la Vision in Paris, which, when associated with the gene of interest, allows the platform to specifically address defects inside the mitochondria using an AAV vector (Adeno-Associated Virus). The gene of interest is transferred into the cell to be expressed and produces the functional protein, which will then be shuttled to the mitochondria through specific nucleotidic sequences in order to restore the missing or deficient mitochondrial function.
About Leber Hereditary Optic Neuropathy (LHON)
Leber Hereditary Optic Neuropathy (LHON) is a rare maternally inherited mitochondrial genetic disease, characterized by the degeneration of retinal ganglion cells that results in brutal and irreversible vision loss that can lead to legal blindness, and mainly affects adolescents and young adults. LHON is associated with painless, sudden loss of central vision in the 1st eye, with the 2nd eye sequentially impaired. It is a symmetric disease with poor functional visual recovery. 97% of patients have bilateral involvement at less than one year of onset of vision loss, and in 25% of cases, vision loss occurs in both eyes simultaneously. The estimated incidence of LHON is approximately 1,400 to 1,500 new patients who lose their sight every year in the United States and Europe.
About RESCUE and REVERSE
RESCUE and REVERSE are two separate randomized, double-masked, sham-controlled Phase III trials designed to evaluate the efficacy of a single intravitreal injection of GS010 (rAAV2/2-ND4) in subjects affected by LHON due to the G11778A mutation in the mitochondrial ND4 gene.
The primary endpoint will measure the difference in efficacy of GS010 in treated eyes compared to sham-treated eyes based on Best‑Corrected Visual Acuity (BCVA), as measured with the ETDRS at 48 weeks post-injection. The patients’ LogMAR (Logarithm of the Minimal Angle of Resolution) scores, which are derived from the number of letters patients read on the ETDRS chart, will be used for statistical purposes. Both trials have been adequately powered to evaluate a clinically relevant difference of at least 15 ETDRS letters between treated and untreated eyes adjusted to baseline.
The secondary endpoints will involve the application of the primary analysis to best‑seeing eyes that received GS010 compared to those receiving sham, and to worse‑seeing eyes that received GS010 compared to those that received sham. Additionally, a categorical evaluation with a responder analysis will be evaluated, including the proportion of patients who maintain vision (< ETDRS 15L loss), the proportion of patients who gain 15 ETDRS letters from baseline and the proportion of patients with Snellen acuity of >20/200. Complementary vision metrics will include automated visual fields, optical coherence tomography, and color and contrast sensitivity, in addition to quality of life scales, bio‑dissemination and the time course of immune response. Readouts for these endpoints are at 48, 72 and 96 weeks after injection.
The trials are conducted in parallel, in 37 subjects for REVERSE and 39 subjects for RESCUE, in 7 centers across the United States, the UK, France, Germany and Italy. Week 96 results were reported in 2019 for both trials, after which patients were transferred to a long-term follow-up study that will last for three years.
ClinicalTrials.gov Identifiers:
REVERSE: NCT02652780
RESCUE: NCT02652767
About REFLECT
REFLECT is a multi-center, randomized, double-masked, placebo-controlled study to evaluate the safety and efficacy of bilateral injections of GS010 in subjects with LHON due to the NADH dehydrogenase 4 (ND4) mutation.
The trial planned to enroll 90 patients with vision loss up to 1 year in duration and will be conducted in multiple centers in Europe and in the US.
In the active arm, GS010 will be administered as a single intravitreal injection to both eyes of each subject. In the placebo arm, GS010 will be administered as a single intravitreal injection to the first affected eye, while the fellow eye will receive a placebo injection.
The primary endpoint for the REFLECT trial is the BCVA reported in LogMAR at 1-Year post-treatment in the second‑affected/not‑yet‑affected eye. The change from baseline in second‑affected/not‑yet‑affected eyes receiving GS010 and placebo will be the primary response of interest. The secondary efficacy endpoints include: BCVA reported in LogMAR at 2-Years post-treatment in the second‑affected/not‑yet‑affected eye compared to both placebo and the first‑affected eye receiving GS010, OCT and contrast sensitivity and quality of life scales. The first subject was treated in March 2018, and enrolment was completed in July 2019, ahead of schedule.
ClinicalTrials.gov Identifiers:
REFLECT: NCT03293524