- Clinically meaningful difference between visual outcomes of LUMEVOQ®-treated patients and untreated patients
Paris, France, June 1, 2021, 7:30 am CEST – GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal neurodegenerative diseases and central nervous system disorders, today announced that the journal Frontiers in Neurology has published results of the indirect comparison of the evolution of visual outcomes in patients treated with LUMEVOQ® gene therapy with the spontaneous evolution in natural history (NH) studies of Leber hereditary optic neuropathy (LHON) patients carrying the m.11778G>A ND4 mutation (MT-ND4 patients).
The paper*, published in the May issue under the title, “Intravitreal Gene Therapy vs. Natural History in Patients with Leber Hereditary Optic Neuropathy Carrying the m.11778G>A ND4 Mutation: Systematic Review and Indirect Comparison”, found a statistically and clinically relevant difference in visual acuities in favor of LUMEVOQ®-treated patients versus untreated NH patients. LUMEVOQ®-treated patients’ best-corrected visual acuity (BCVA) experienced progressive and sustained improvement from Month 12 to Month 52 after vision loss, whereas NH patients showed deteriorating visual acuity over the same time period.
Figure 1. Evolution of visual acuities of treated eyes vs. natural history eyes
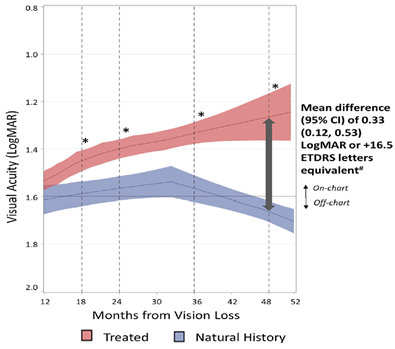
Note: All patients had a confirmed G11778A mutation in the ND4 mitochondrial gene and were at least 15 years old at onset. The diagram shows the Locally Estimated Scatterplot Smoothing (LOESS) curves for visual acuity in LUMEVOQ®-treated patients and untreated patients, with the shaded areas representing the 95% confidence interval for the mean BCVA. “Treated” eyes refer to all eyes (LUMEVOQ® and sham) from the RESCUE, REVERSE and CLIN06 trials (N=152 eyes from 76 patients). Untreated eyes refer to patient-level data from a matched data set from the REALITY study, two prospective and eight retrospective natural history studiesd (N=408 eyes from 208 patients). *A statistically significant difference between treated and natural history eyes is illustrated by the non-overlapping confidence intervals of the LOESS curves.
“Detailed analysis of what the literature can tell us about the natural history of this devastating blinding disorder does confirm its poor visual prognosis,” said Dr. Nancy J. Newman, MD, lead and corresponding author, RESCUE principal investigator and LeoDelle Jolley Professor of Ophthalmology and Neurology at the Emory University School of Medicine in Atlanta, GA, USA.
The publication represents a peer-review validation of the analyses submitted in LUMEVOQ®’s Marketing Authorisation Application in Europe, which was filed in September 2020. Review of the dossier is ongoing, with the EMA decision expected in H1 2022.
Overview of the indirect comparison methodology
The indirect comparison approach was pursued because an unexpected and significant contralateral effect in the RESCUEa and REVERSEb Phase III trials effectively eliminated the sham-injected eyes as a control group for the drug-injected eyes. For a better characterization of LUMEVOQ® efficacy, a natural history, or untreated, pool had to be assembled from patients carrying the same mutation as those in the clinical trials.
The authors formed the “efficacy pool” by combining all visual acuity observations from inclusion to Week 96 in the 76 RESCUE and REVERSE patients, with those of patients who accepted to participate in the long-term follow-up study RESTORE (CLIN06). Because of the contralateral effect, visual acuity measurements for sham-treated eyes were included in the efficacy dataset.
The natural history pool was formed from patients who were not treated with LUMEVOQ®; they could, however, have been treated with idebenone. The dataset was created using visual outcome data from the REALITY LHON registryc and 10 published studies on LHON that were identified after a systematic review of the literatured. Only studies with patient- and eye-level visual acuity values, along with documentation of the time after vision loss in cohorts with at least five MT-ND4 patients, were included (208 patients).
Month 12 after vision loss was selected as the starting point of the indirect comparison, as nearly all (93%) of the eyes in the efficacy pool had been treated at that point. The evolution of the visual acuity was estimated using a locally estimated scatterplot smoothing (LOESS), non-parametric, local regression model.
Key findings from the indirect comparison
Treated eyes had better visual acuity compared to natural history eyes at 12, 18, 24, 36 and 48 months after vision loss. The difference in mean visual acuity was statistically significant at all time points, with the clinically meaningful difference at 48 months amounting to +16.5 ETDRS letters equivalent (more than +3 lines on the ETDRS vision chart) in favor of treated eyes. The treatment effect was confirmed even after confounding covariates, such as age at onset of vision loss, gender and duration of follow-up, were taken into account using multivariate analysis.
The treatment effect was similarly reflected in eye responder rates, which was approximately double when patients were treated, regardless of the responder measure selected (e.g., percentage of eyes with on-chart visual acuity at Month 48 and at last observation; percentage of eyes above the WHO criteria for blindness at Month 48 and at last observation).
The paper can be obtained at https://www.frontiersin.org/articles/10.3389/fneur.2021.662838/full.
*About the paper:
Intravitreal Gene Therapy vs. Natural History in Patients with Leber Hereditary Optic Neuropathy Carrying the m.11778G>A ND4 Mutation: Systematic Review and Indirect Comparison
Authors: Nancy J. Newman1, Patrick Yu-Wai-Man,2,3,4,5, Valerio Carelli6,7, Valerie Biousse1, Mark L. Moster8, Catherine Vignal-Clermont9,10, Robert C. Sergott8, Thomas Klopstock11,12,13, Alfredo A. Sadun14, Jean François Girmens10, Chiara La Morgia6, Adam A. DeBusk8, Neringa Jurkute4,5, Claudia Priglinger15, Rustum Karanjia14,16, Constant Josse17, Julie Saltzmann18, François Montestruc17, Michel Roux19, Magali Taiel19, and José-Alain Sahel 20,21,22,23 for the LHON Study Group
Affiliations:
1 Departments of Ophthalmology, Neurology and Neurological Surgery, Emory University School of Medicine, Atlanta, GA, United States
2 Cambridge Centre for Brain Repair and MRC Mitochondrial Biology Unit, Department of Clinical Neurosciences, University of Cambridge, Cambridge, United Kingdom
3 Cambridge Eye Unit, Addenbrooke’s Hospital, Cambridge University Hospitals, Cambridge, United Kingdom
4 Moorfields Eye Hospital National Health Service Foundation Trust, London, United Kingdom
5 UCL Institute of Ophthalmology, University College London, London, United Kingdom
6 Istituto di Recovero e Cura a Carattere Scientifico Istituto delle Scienze Neurologiche di Bologna, Unità Operativa Compless Clinica Neurologica, Bologna, Italy
7 Unit of Neurology, Department of Biomedical and Neuromotor Sciences, University of Bologna, Bologna, Italy
8 Departments of Neurology and Ophthalmology, Wills Eye Hospital and Thomas Jefferson University, Philadelphia, PA, United States
9 Department of Neuro Ophthalmology and Emergencies, A. de Rothschild Foundation Hospital, Paris, France
10 Centre d’investigation Clinique, Centre Hospitalier National d’Ophtalmologie des Quinze Vingts, Paris, France
11 Department of Neurology, Friedrich-Baur-Institute, University Hospital, Ludwig-Maximilians-University Munich, Munich, Germany
12 German Center for Neurodegenerative Diseases, Munich, Germany
13 Munich Cluster for Systems Neurology, Munich, Germany
14 Doheny Eye Institute, University of California School of Medicine, Los Angeles, CA, United States
15 Department of Ophthalmology, University Hospital, Ludwig-Maximilians-University Munich, Munich, Germany
16 Department of Ophthalmology, University of Ottawa Eye, Ottawa, ON, Canada
17 eXYSTAT, Data Management and Statistic, Malakoff, France
18 Medical and Regulatory Consulting, Paris, France
19 GenSight Biologics, Paris, France
20 Sorbonne Université, INSERM, CNRS, Institut de la Vision, Paris, France
21 A. de Rothschild Foundation Hospital, Paris, France
22 Department of Ophthalmology, The University of Pittsburgh School of Medicine, Pittsburgh, PA, United States
23 CHNO des Quinze-Vingts, Institut Hospitalo-Universitaire FOReSIGHT, INSERM-DGOS CIC 1423, Paris, France
Notes:
a Newman NJ, Yu-Wai-Man P, Carelli V, Moster ML, Biousse V, Vignal-Clermont C, et al. Efficacy and safety of intravitreal gene therapy for leber hereditary optic neuropathy treated within 6 months of disease onset. Ophthalmology. (2021) 128:649–60. doi: 10.1016/j.ophtha.2020.12.012.
b Yu-Wai-Man P, Newman NJ, Carelli V, Moster ML, Biousse V, Sadun AA, et al. Bilateral visual improvement with unilateral gene therapy injection for Leber hereditary optic neuropathy. Sci Transl Med. (2020) 12:eaaz7423. doi: 10.1126/scitranslmed.aaz7423
c Yu-Wai-Man P, Newman NJ, Carelli V, La Morgia C, Biousse V, Bandello FM, et al. Natural history of patients with Leber hereditary optic neuropathy—results from the REALITY study. Eye (2021). https://doi.org/10.1038/s41433-021-01535-9
d Newman NJ, Carelli V, Taiel M, Yu-Wai-Man P. Visual outcomes in Leber hereditary optic neuropathy patients with the m.11778G>A (MTND4) mitochondrial dna mutation. J Neuroophthalmol. (2020) 40:547–57. doi: 10.1097/WNO.0000000000001045.
About GenSight Biologics
GenSight Biologics S.A. is a clinical-stage biopharma company focused on developing and commercializing innovative gene therapies for retinal neurodegenerative diseases and central nervous system disorders. GenSight Biologics’ pipeline leverages two core technology platforms, the Mitochondrial Targeting Sequence (MTS) and optogenetics, to help preserve or restore vision in patients suffering from blinding retinal diseases. GenSight Biologics’ lead product candidate, LUMEVOQ® (GS010; lenadogene nolparvovec), has been submitted for marketing approval in Europe for the treatment of Leber Hereditary Optic Neuropathy (LHON), a rare mitochondrial disease affecting primarily teens and young adults that leads to irreversible blindness. Using its gene therapy-based approach, GenSight Biologics’ product candidates are designed to be administered in a single treatment to each eye by intravitreal injection to offer patients a sustainable functional visual recovery.
About Leber Hereditary Optic Neuropathy (LHON)
Leber Hereditary Optic Neuropathy (LHON) is a rare maternally inherited mitochondrial genetic disease, characterized by the degeneration of retinal ganglion cells that results in brutal and irreversible vision loss that can lead to legal blindness, and mainly affects adolescents and young adults. LHON is associated with painless, sudden loss of central vision in the 1st eye, with the 2nd eye sequentially impaired. It is a symmetric disease with poor functional visual recovery. 97% of patients have bilateral involvement at less than one year of onset of vision loss, and in 25% of cases, vision loss occurs in both eyes simultaneously. The estimated incidence of LHON is approximately 800-1,200 new patients who lose their sight every year in the United States and the European Union.
About LUMEVOQ® (GS010; lenadogene nolparvovec)
LUMEVOQ® (GS010; lenadogene nolparvovec) targets Leber Hereditary Optic Neuropathy (LHON) by leveraging a mitochondrial targeting sequence (MTS) proprietary technology platform, arising from research conducted at the Institut de la Vision in Paris, which, when associated with the gene of interest, allows the platform to specifically address defects inside the mitochondria using an AAV vector (Adeno-Associated Virus). The gene of interest is transferred into the cell to be expressed and produces the functional protein, which will then be shuttled to the mitochondria through specific nucleotidic sequences in order to restore the missing or deficient mitochondrial function. “LUMEVOQ” was accepted as the invented name for GS010 (lenadogene nolparvovec) by the European Medicines Agency (EMA) in October 2018.
About RESCUE and REVERSE
RESCUE and REVERSE are two separate randomized, double-masked, sham-controlled Phase III trials designed to evaluate the efficacy of a single intravitreal injection of GS010 (rAAV2/2-ND4) in subjects affected by LHON due to the G11778A mutation in the mitochondrial ND4 gene.
The primary endpoint measured the difference in efficacy of GS010 in treated eyes compared to sham-treated eyes based on Best‑Corrected Visual Acuity (BCVA), as measured with the ETDRS at 48 weeks post-injection. The patients’ LogMAR (Logarithm of the Minimal Angle of Resolution) scores, which are derived from the number of letters patients read on the ETDRS chart, was used for statistical purposes. Both trials were adequately powered to evaluate a clinically relevant difference of at least 15 ETDRS letters between treated and untreated eyes adjusted to baseline.
The secondary endpoints involved the application of the primary analysis to best‑seeing eyes that received GS010 compared to those receiving sham, and to worse‑seeing eyes that received GS010 compared to those that received sham. Additionally, a categorical evaluation with a responder analysis was evaluated, including the proportion of patients who maintain vision (< ETDRS 15L loss), the proportion of patients who gain 15 ETDRS letters from baseline and the proportion of patients with Snellen acuity of >20/200. Complementary vision metrics included automated visual fields, optical coherence tomography, and color and contrast sensitivity, in addition to quality of life scales, bio‑dissemination and the time course of immune response. Readouts for these endpoints were at 48, 72 and 96 weeks after injection.
The trials were conducted in parallel, in 37 subjects for REVERSE and 39 subjects for RESCUE, in 7 centers across the United States, the UK, France, Germany and Italy. Week 96 results were reported in 2019 for both trials, after which patients were invited to a long-term follow-up study that will last for three years.
ClinicalTrials.gov Identifiers:
REVERSE: NCT02652780
RESCUE: NCT02652767
About REALITY
REALITY is a multi-country retrospective and cross-sectional observational study of affected LHON subjects, based on subjects’ medical charts and the administration of surveys on Health-Related Quality of Life (HRQoL) and direct and indirect costs associated with the disease. The study enrolled 44 subjects (both adult and pediatric) chiefly in the following countries: Spain, Italy, France, United Kingdom, and the United States.
The primary objective for the REALITY study was to describe the evolution of visual functional and structural changes and other associated symptoms in patients with LHON; understand the impact of LHON-related vision loss on the HRQoL; and understand the economic burden for patients and their families arising from direct and indirect costs associated with the disease. The secondary objective is to describe the relationship between genetic, lifestyle and/or environmental factors and the expression of the LHON phenotype.
The first subject was enrolled on 3 January 2018, and enrollment was completed in early Q2 2020.
ClinicalTrials.gov Identifiers:
REALITY LHON Registry: NCT03295071
Contacts
-
LifeSci AdvisorsInvestor RelationsGuillaume van Renterghem+41 (0)76 735 01 31