- Treatment effect of LUMEVOQ® was maintained at Year 3 after treatment administration, with mean visual acuity achieving an on-chart level of 1.26 LogMAR
- Quality of life scores continued to improve between Year 2 and Year 3 after treatment administration
Paris, France, August 31, 2021, 7:30 am CET – GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal neurodegenerative diseases and central nervous system disorders, today announced that the Journal of Neuro-Ophthalmology (JNO) has published results from RESTORE, the long-term follow-up study of LUMEVOQ®, which show sustained treatment effect from a unilateral injection of LUMEVOQ® three years after injection in the RESCUE and REVERSE trials.
The paper*, published in the September issue of JNO under the title “Long-Term Follow-Up After Unilateral Intravitreal Gene Therapy for Leber Hereditary Optic Neuropathy: The RESTORE Study”, presents analyses that show sustained improvement in best-corrected visual acuity (BCVA) and quality of life scores three years after subjects received LUMEVOQ® treatment. The continuous improvement in BCVA was demonstrated in both eyes of the unilaterally treated patients, confirming the contralateral treatment effect reported in the RESCUE and REVERSE trials.
“It is gratifying to see this sustained outcome”, commented lead author Dr. Valérie Biousse, MD, Departments of Ophthalmology and Neurology, Emory University School of Medicine, Atlanta, GA. Dr. Biousse, who was also an co‑investigator in the RESCUE and REVERSE trials, added, “This is further evidence of a bilateral therapeutic effect of a single unilateral gene therapy injection.”
Mean BCVA steadily improved to 1.26 LogMAR at 48 months after onset (3 year-post injection), remaining on‑chart (i.e., better than 1.6 LogMAR) throughout the follow-up period. A locally-estimated scatterplot smoothing (LOESS) regression analysis illustrates the progressive and sustained improvement of BCVA in RESTORE subjects (Figure 1) since treatment with LUMEVOQ®.
Figure 1. Evolution of best-corrected visual acuity (BCVA) in ND4-LHON subjects who received unilateral injection of LUMEVOQ® gene therapy
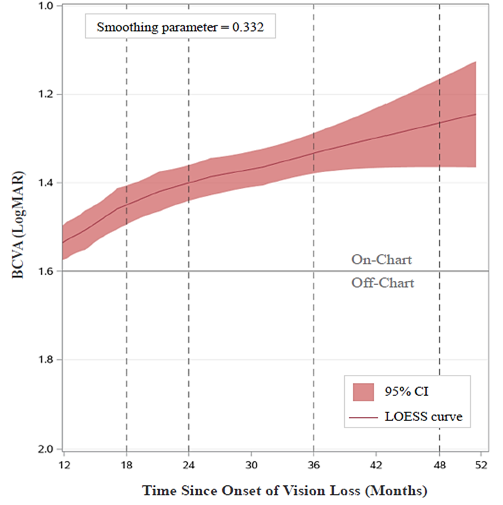
BCVA: best-corrected visual acuity, CI: confidence interval; LOESS: locally estimated scatterplot smoothing, LogMAR: logarithm of the minimal angle resolution. The LOESS curve shows the evolution of BCVA in all eyes (LUMEVOQ®– and sham-treated) from 12 months to 51.5 months (last available data point) after onset of vision loss. The curve starts at 12 months after onset when 92.7% of eyes in RESCUE and REVERSE had received treatment, either with LUMEVOQ® or a sham injection.
In addition, subjects’ quality of life continued to improve between Year 2 and Year 3 post-injection, as documented by scores reported in the visual function questionnaire VFQ-25. Relative to baseline, the mean VFQ-25 composite score (averaging 11 vision‑related subscales) was higher by 4 points at Year 2 and 7 points at Year 3. At Year 3, clinically meaningful improvement from baseline were seen in the sub-scores that corresponded to mental health (+21 points), role difficulties (+17 points), dependency (+15 points), general vision (+9 points), near activities (+6 points), and distance activities (+5 points).
RESCUE and REVERSE were randomized, double-masked, sham-controlled, Phase III clinical trials that assessed the efficacy and safety of LUMEVOQ® gene therapy as a treatment for vision loss due to ND4-LHON. The only difference between the two studies was the duration of vision loss at screening: RESCUE subjects had vision loss for less than 6 months, while REVERSE subjects had vision loss for 6 to 12 months. The 72 subjects who completed the Phase III trials RESCUE and REVERSE were invited to participate in RESTORE, and 62 (86.1%) agreed to be monitored up to five years after treatment.
The paper is available at: https://journals.lww.com/jneuro-ophthalmology/Fulltext/2021/09000/Long_Term_Follow_Up_After_Unilateral_Intravitreal.5.aspx.
*About the paper:
Long-Term Follow-Up After Unilateral Intravitreal Gene Therapy for Leber Hereditary Optic Neuropathy: The RESTORE Study
Authors: Valérie Biousse, MD1, Nancy J. Newman, MD1, Patrick Yu-Wai-Man, MD, PhD2,3,4,5, Valerio Carelli, MD PhD6,7 , Mark L. Moster, MD8, Catherine Vignal-Clermont, MD9,10, Thomas Klopstock, MD11,12,13, Alfredo A. Sadun, MD, PhD14, Robert C. Sergott, MD8, Rabih Hage, MD10, Simona Esposti, MD4, Chiara La Morgia, MD, PhD6,7, Claudia Priglinger, MD15, Rustum Karanja, MD, PhD14,16, Laure Blouin, MSc17, Magali Taiel, MD17, José-Alain Sahel, MD, PhD10,18,19,20 for the LHON Study Group
Affiliations:
- Departments of Ophthalmology, Neurology and Neurological Surgery, Emory University School of Medicine, Atlanta, Georgia USA.
- Cambridge Centre for Brain Repair and MRC Mitochondrial Biology Unit, Department of Clinical Neurosciences, University of Cambridge, Cambridge, UK.
- Cambridge Eye Unit, Addenbrooke’s Hospital, Cambridge University Hospitals, Cambridge, UK.
- Moorfields Eye Hospital, London, UK.
- UCL Institute of Ophthalmology, University College London, London, UK.
- IRCCS Istituto delle Scienze Neurologiche di Bologna, Programma di Neurogenetica, Bologna, Italy.
- Unit of Neurology, Department of Biomedical and Neuromotor Sciences (DIBINEM), University of Bologna, Bologna, Italy.
- Departments of Neurology and Ophthalmology, Wills Eye Hospital and Thomas Jefferson University, Philadelphia, PA, USA.
- Department of Neuro Ophthalmology and Emergencies, Rothschild Foundation Hospital, Paris, France.
- Centre Hospitalier National d’Ophtalmologie des Quinze Vingts, Institut Hospitalo-Universitaire FOReSIGHT, INSERM-DGOS CIC 1423, Paris, France
- Department of Neurology, Friedrich-Baur-Institute, University Hospital, LMU Munich, 80336 Munich, Germany.
- German Center for Neurodegenerative Diseases (DZNE), Munich, Germany.
- Munich Cluster for Systems Neurology (SyNergy), Munich, Germany.
- Doheny Eye Institute / UCLA School of Medicine, Los Angeles, CA, USA
- Department of Ophthalmology, University Hospital, Ludwig-Maximilians-University (LMU) Munich, 80336 Munich, Germany
- Department of Ophthalmology, University of Ottawa Eye, Ottawa ON, Canada
- GenSight Biologics, Paris, France
- Sorbonne Université, INSERM, CNRS, Institut de la Vision, 75012 Paris, France
- Fondation Ophtalmologique A. de Rothschild, 25-29 rue Manin, 75019 Paris
- Department of Ophthalmology, The University of Pittsburgh School of Medicine, Pittsburgh, USA
About GenSight Biologics
GenSight Biologics S.A. is a clinical-stage biopharma company focused on developing and commercializing innovative gene therapies for retinal neurodegenerative diseases and central nervous system disorders. GenSight Biologics’ pipeline leverages two core technology platforms, the Mitochondrial Targeting Sequence (MTS) and optogenetics, to help preserve or restore vision in patients suffering from blinding retinal diseases. GenSight Biologics’ lead product candidate, LUMEVOQ® (GS010; lenadogene nolparvovec), has been submitted for marketing approval in Europe for the treatment of Leber Hereditary Optic Neuropathy (LHON), a rare mitochondrial disease affecting primarily teens and young adults that leads to irreversible blindness. Using its gene therapy-based approach, GenSight Biologics’ product candidates are designed to be administered in a single treatment to each eye by intravitreal injection to offer patients a sustainable functional visual recovery.
About Leber Hereditary Optic Neuropathy (LHON)
Leber Hereditary Optic Neuropathy (LHON) is a rare maternally inherited mitochondrial genetic disease, characterized by the degeneration of retinal ganglion cells that results in brutal and irreversible vision loss that can lead to legal blindness, and mainly affects adolescents and young adults. LHON is associated with painless, sudden loss of central vision in the 1st eye, with the 2nd eye sequentially impaired. It is a symmetric disease with poor functional visual recovery. 97% of patients have bilateral involvement at less than one year of onset of vision loss, and in 25% of cases, vision loss occurs in both eyes simultaneously. The estimated incidence of LHON is approximately 800-1,200 new patients who lose their sight every year in the United States and the European Union.
About LUMEVOQ® (GS010; lenadogene nolparvovec)
LUMEVOQ® (GS010; lenadogene nolparvovec) targets Leber Hereditary Optic Neuropathy (LHON) by leveraging a mitochondrial targeting sequence (MTS) proprietary technology platform, arising from research conducted at the Institut de la Vision in Paris, which, when associated with the gene of interest, allows the platform to specifically address defects inside the mitochondria using an AAV vector (Adeno-Associated Virus). The gene of interest is transferred into the cell to be expressed and produces the functional protein, which will then be shuttled to the mitochondria through specific nucleotidic sequences in order to restore the missing or deficient mitochondrial function. “LUMEVOQ” was accepted as the invented name for GS010 (lenadogene nolparvovec) by the European Medicines Agency (EMA) in October 2018.
About RESCUE, REVERSE, and RESTORE
RESCUE and REVERSE were two separate randomized, double-masked, sham-controlled Phase III trials designed to evaluate the efficacy of a single intravitreal injection of GS010 (rAAV2/2-ND4) in subjects affected by LHON due to the G11778A mutation in the mitochondrial ND4 gene.
The primary endpoint measured the difference in efficacy of GS010 in treated eyes compared to sham-treated eyes based on Best‑Corrected Visual Acuity (BCVA), as measured with the ETDRS at 48 weeks post-injection. The patients’ LogMAR (Logarithm of the Minimal Angle of Resolution) scores, which are derived from the number of letters patients read on the ETDRS chart, were used for statistical purposes. Both trials were adequately powered to evaluate a clinically relevant difference of at least 15 ETDRS letters between drug-treated and sham-treated eyes, adjusted to baseline.
The secondary endpoints involved the application of the primary analysis to best‑seeing eyes that received GS010 compared to those receiving sham, and to worse‑seeing eyes that received GS010 compared to those that received sham. Additionally, a categorical evaluation with a responder analysis was performed, including the proportion of patients who maintained vision (< ETDRS 15L loss), the proportion of patients who gained 15 ETDRS letters from baseline and the proportion of patients with Snellen acuity of >20/200. Complementary vision metrics included automated visual fields, optical coherence tomography, and color and contrast sensitivity, in addition to quality-of-life scales, bio‑dissemination and the time course of immune response. Readouts for these endpoints were at 48, 72 and 96 weeks after injection.
The trials were conducted in parallel, in 37 subjects for REVERSE and 39 subjects for RESCUE, in 7 centers across the United States, the UK, France, Germany and Italy. Week 96 results were reported in 2019 for both trials, after which patients were invited to participate in a long-term follow-up study, RESTORE, for three additional years.
The primary objective is to assess the long-term safety of intravitreal LUMEVOQ® administration up to 5 years post-treatment. The secondary objective is to assess the long-term treatment efficacy of the therapy and the quality of life (QoL) in subjects up to 5 years post-treatment. The first subject was enrolled on January 9, 2018. 61 subjects have enrolled.
ClinicalTrials.gov Identifiers:
REVERSE: NCT02652780
RESCUE: NCT02652767
RESTORE: NCT03406104
Contacts
-
LifeSci AdvisorsInvestor RelationsGuillaume van Renterghem+41 (0)76 735 01 31