- Sustained efficacy 4 years after injection in REFLECT Phase III trial
- Continued benefit at 4 years in bilaterally treated patients: clinically meaningful improvement of at least 15 letters relative to their observed nadir
- 73% of bilaterally treated patients experience clinically meaningful level of improvement
- Favorable safety profile confirmed at 4 years
Paris, France, March 20, 2024, 7:30 am CET – GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal neurodegenerative diseases and central nervous system disorders, today reported initial efficacy and safety results at 4 years[1] post-treatment administration in the REFLECT Phase III clinical trial with LUMEVOQ® (GS010; lenadogene nolparvovec). The results show that four years after a one-time administration of the gene therapy, the visual acuity improvement has been sustained while maintaining a favorable safety profile. Bilateral injection provides an additional effect compared to unilateral treatment, demonstrated across all analyses of visual acuity improvement and responder rates.
“The latest REFLECT data confirms that the improvement seen with lenadogene nolparvovec is sustained 4 years after treatment has been given, including the additional benefit observed in participants receiving a bilateral intravitreal injection of the gene therapy,” said Patrick Yu-Wai-Man, MD, PhD, Professor of Ophthalmology, University of Cambridge and Moorfields Eye Hospital, and International Principal Investigator of REFLECT. “Importantly, REFLECT participants receiving a bilateral injection had a comparable safety profile to those treated unilaterally.”
The findings reinforce the results observed at 3 years post-treatment administration, which were reported in March 2023.[2]
Sustained and meaningful efficacy at Year 4
The evolution of the visual acuity over time shows that visual improvement after lenadogene nolparvovec treatment was maintained over 4 years in all subjects, with the visual acuity of bilateral injected patients remaining better than that of patients who received a unilateral injection. This difference has been observed since Year 1.5.
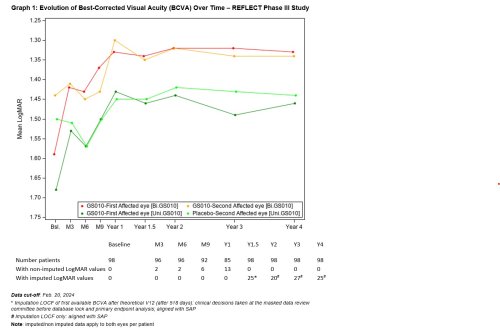
Compared against nadir (i.e., the worst BCVA recorded from baseline to Year 4), average visual acuity for all LUMEVOQ-treated eyes increased beyond the +15 letter threshold that conventionally defines clinically meaningful improvement. The improvement of placebo eyes highlights the consistent contralateral treatment effect observed in all clinical trials (also documented in sham-treated eyes in the REVERSE1 and RESCUE2 trials)
“The sustained effect on vision, as observed in the REFLECT trial, is a crucial piece of the LUMEVOQ story for patients, physicians and health authorities,” commented Laurence Rodriguez, Chief Executive Officer of GenSight Biologics. “The durable impact from a single administration differentiates gene therapy from other treatment modalities, facilitating patient adherence and improving quality of life.”
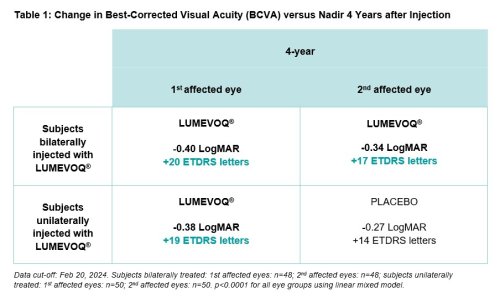
Responder analyses reinforce the finding of improved outcomes for patients, for whom natural history typically results in greatly impaired vision with a very low likelihood of spontaneous recovery.3 Four years after a bilateral injection, 73% of patients had experienced a clinically meaningful improvement of at least -0.3 LogMAR (+15 ETDRS letters) relative to their observed nadir. 81% of bilaterally treated patients are able to read letters on a screen (on-chart vision), with the likelihood of reaching this level of vision being twice as high with bilateral treatment versus a unilateral injection (odds ratio: 2.0 [0.7; 5.5]).
Favorable safety profile
The favorable safety profile of LUMEVOQ® continued to be confirmed, with the safety profile of the drug being demonstrated as comparable in bilaterally and unilaterally treated subjects. There was no study discontinuation related to systemic or ocular adverse events, and there were no serious ocular adverse events. The main ocular adverse event was intraocular inflammation, which was mostly mild and responsive to conventional treatment.
REFLECT is a randomized, double-masked, placebo-controlled Phase III trial involving 98 subjects with vision loss due to Leber Hereditary Optic Neuropathy (LHON) caused by a mutated ND4 mitochondrial gene; enrolled ND4 subjects had vision loss up to one year from onset. All subjects received an intravitreal injection (IVT) of lenadogene nolparvovec in their first affected eye. The second affected eye was randomized to either a second IVT of LUMEVOQ® or a placebo IVT, which was administered on the same day or the following day. 48 subjects were randomized to LUMEVOQ® bilateral treatment, and 50 to lenadogene nolparvovec unilateral treatment (first-affected eye treated with LUMEVOQ®, second-affected eye treated with placebo). REFLECT patients will be followed up to 5 years post-injection to monitor the efficacy and safety of LUMEVOQ® over time.
References:
- Yu-Wai-Man P, Newman NJ, Carelli V, Moster ML, Biousse V, Sadun AA, et al. Bilateral visual improvement with unilateral gene therapy injection for Leber hereditary optic neuropathy. Sci Transl Med. (2020) 12:eaaz7423. doi: 10.1126/scitranslmed.aaz7423
- Newman NJ, Yu-Wai-Man P, Carelli V, Moster ML, Biousse V, Vignal-Clermont C, et al. Efficacy and safety of intravitreal gene therapy for Leber hereditary optic neuropathy treated within 6 months of disease onset. Ophthalmology (2021) 128:649–60. doi: 10.1016/j.ophtha.2020.12.012.
- Newman NJ, Carelli V, Taiel M, Yu-Wai-Man P. Visual outcomes in Leber hereditary optic neuropathy subjects with the m.11778G>A (MTND4) mitochondrial DNA mutation. J Neuro-Ohthalmol. (2020) 40:547–57. doi: 10.1097/WNO.0000000000001045.
[1] Data cut-off: February 20, 2024
[2] “GenSight Biologics Confirms Sustained Efficacy and Safety of Bilateral LUMEVOQ® Injections at 3-Year Follow-Up of REFLECT Phase III Trial”, press release, March 13, 2023
Contacts
-
LifeSci AdvisorsInvestor RelationsGuillaume van Renterghem+41 (0)76 735 01 31