- Sustained efficacy three years after injection: statistically significant visual acuity improvement from nadir
- Continuous benefits in bilaterally treated patients with +17 to +20 ETDRS letters improvement vs nadir at 3 years
- 62% of bilaterally treated patients off-chart at baseline move back to on-chart territory at 3 years
- Good safety profile confirmed at 3 years
Paris, France, March 13, 2023, 7:30 am CET – GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal neurodegenerative diseases and central nervous system disorders, today reported topline efficacy and safety results at 3 years[1] post-treatment administration in the REFLECT Phase III clinical trial with LUMEVOQ® (lenadogene nolparvovec). The results show sustained efficacy and favorable safety for bilateral intravitreal injection of the gene therapy with a statistically significant visual acuity improvement from baseline in both treated eyes, showing an additional benefit of a bilateral injection compared to a unilateral injection.
“The REFLECT trial has delivered a further set of results demonstrating the sustained benefit of lenadogene nolparvovec, while reconfirming its safety profile ” said Nancy J Newman, MD, Professor of Ophthalmology and Neurology at Emory University School of Medicine (Atlanta, USA) and International Principal Investigator of REFLECT. “ In addition, the additional benefit among bilaterally treated patients suggests that injection of both eyes may be the best option for LHON patients with the ND4 mutation.”
The findings reinforce the results observed at 2 years post-treatment administration, which were reported in December 2021, and at 1.5 years post-treatment administration, which were reported in June 2021.
Designed under a Special Protocol Assessment with the US FDA, REFLECT is a randomized, double-masked, placebo-controlled Phase III trial involving 98 subjects with vision loss due to Leber Hereditary Optic Neuropathy (LHON) caused by a mutated ND4 mitochondrial gene; enrolled ND4 subjects had vision loss up to one year from onset. Vision typically deteriorates to a nadir and stabilises to a level below the threshold for legal blindness. The ND4 mitochondrial mutation is associated with the most severe clinical form of LHON, with poor overall visual outcomes.1 All subjects received an intravitreal injection (IVT) of lenadogene nolparvovec in their first affected eye. The second affected eye was randomized to either a second IVT of LUMEVOQ® or a placebo IVT, which was administered on the same day or the following day. 48 subjects were randomized to LUMEVOQ® bilateral treatment, and 50 to lenadogene nolparvovec unilateral treatment (first-affected eye treated with lenadogene nolparvovec, second-affected eye treated with placebo).
Sustained and meaningful efficacy at year 3
The evolution of the visual acuity over time shows that lenadogene nolparvovec effect is maintained over 3 years for all subjects, with an additional benefit with a bilateral injection as compared to a unilateral injection. Year 3 analyses confirm the dose effect that was noted at Year 1.5 and Year 2, with an absolute difference of the mean visual acuity between arms of +6,5 ETDRS letters in favor of bilaterally treated subjects at Year 3.
Graph 1: Evolution of Best-Corrected Visual Acuity (BCVA) over time
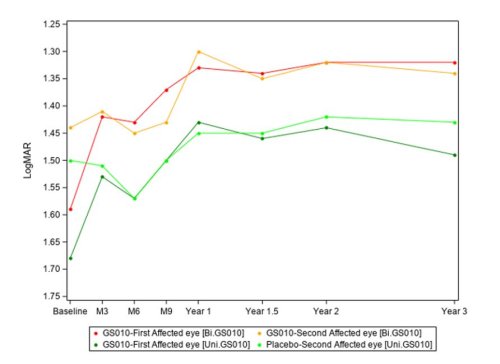
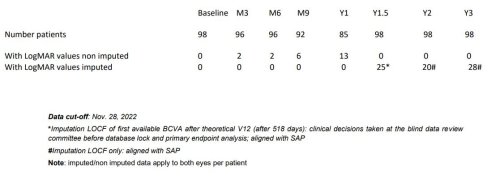
Table 1: Change in Best-Corrected Visual Acuity (BCVA) versus Nadir 3 Years after Injection
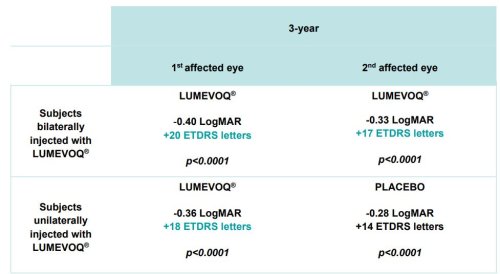
Data cut-off: Nov 28, 2022. Subjects bilaterally treated : 1st affected eyes: n=48; 2nd affected eyes: n=48; subjects unilaterally treated: 1st affected eyes: n=50; 2nd affected eyes: n=50.
Comparison against nadir (i.e., the worst BCVA recorded from baseline to Year 3) starkly demonstrates the efficacy of lenadogene nolparvovec, even for the placebo eyes that improved via a contralateral treatment effect (also documented in sham-treated eyes in the REVERSE2 and RESCUE3 trials).
Responder analyses point to the benefits of treatment for patients that would otherwise have experienced significant vision loss with a very low likelihood of spontaneous recovery (partial recovery in 11% of patients described in the literature).1 Three years after a bilateral injection, 73% of patients had experienced a clinically meaningful improvement of at least -0.3 LogMAR (+15 ETDRS letters) relative to their observed nadir, and, among patients who were not able to read any letters on a screen at baseline (off-chart values for both eyes), 62% are now able to read letters.
Favorable safety profile
The favorable safety profile of lenadogene nolparvovec continued to be confirmed, with the safety profile of the drug being demonstrated as comparable in bilaterally and unilaterally treated subjects. There was no study discontinuation related to systemic or ocular adverse events, and there were no serious ocular adverse events. The main ocular adverse event was intraocular inflammation, which was mostly mild and responsive to conventional treatment.
REFLECT patients will be followed up to 5 years post-injection to monitor the efficacy and safety of lenadogene nolparvovec over time.
“We are very pleased to see that this 3 years’ follow-up results from the REFLECT trial confirm the durable and meaningful efficacy of lenadogene nolparvovec, particularly for bilaterally treated patients,” said Bernard Gilly, CEO and Co-Founder of GenSight Biologics. “The body of clinical evidence is making a compelling case that this gene therapy represents real hope for patients affected by this devastating blinding disease. These latest data will provide further significant support for the ongoing review of our dossier by the EMA.”
References:
- Newman NJ, Carelli V, Taiel M, Yu-Wai-Man P. Visual outcomes in Leber hereditary optic neuropathy subjects with the m.11778G>A (MTND4) mitochondrial dna mutation. J Neuroophthalmol. (2020) 40:547–57. doi: 10.1097/WNO.0000000000001045.
- Yu-Wai-Man P, Newman NJ, Carelli V, Moster ML, Biousse V, Sadun AA, et al. Bilateral visual improvement with unilateral gene therapy injection for Leber hereditary optic neuropathy. Sci Transl Med. (2020) 12:eaaz7423. doi: 10.1126/scitranslmed.aaz7423
- Newman NJ, Yu-Wai-Man P, Carelli V, Moster ML, Biousse V, Vignal-Clermont C, et al. Efficacy and safety of intravitreal gene therapy for Leber hereditary optic neuropathy treated within 6 months of disease onset. Ophthalmology. (2021) 128:649–60. doi: 10.1016/j.ophtha.2020.12.012.
Contacts
-
LifeSci AdvisorsInvestor RelationsGuillaume van Renterghem+41 (0)76 735 01 31